| Lane Lab |
|
Phenology patterns for three mammalian study systems
Phenologies represent the seasonal timings of life history events, and humankind's interest in their expression was likely borne out of necessity. Early hunter-gatherers needed to be able to identify cues to upcoming availabilities of traditional food sources. Whilst our reliance on such cues has lessened in most modern societies, out interests have been maintained. Phenological datasets represent some of the longest running of all biological records (for example, the blossoming of cherry trees in Kyoto, Japan has been diligently observed and reported for more than a millennium). These datasets are now proving to be highly valuable as we attempt to understand the influences of climate change on wild populations. Shifts in phenologies are the most commonly reported ecological consequences of climate change and, of concern, not all components of given ecosystems exhibit synchronous adjustments. Many populations are thus becoming desynchronized from their primary food resources, and population-level declines have been reported.
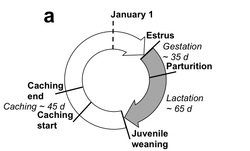
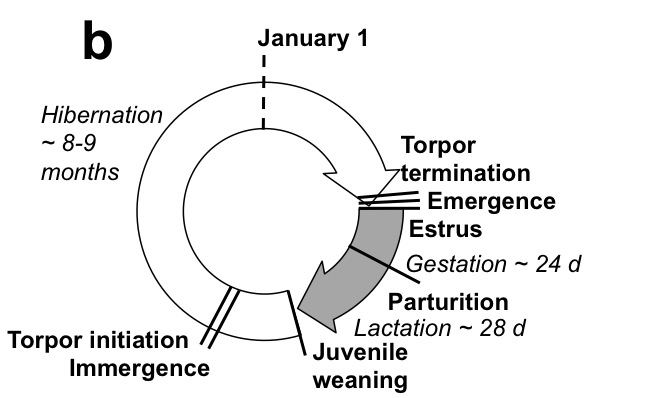
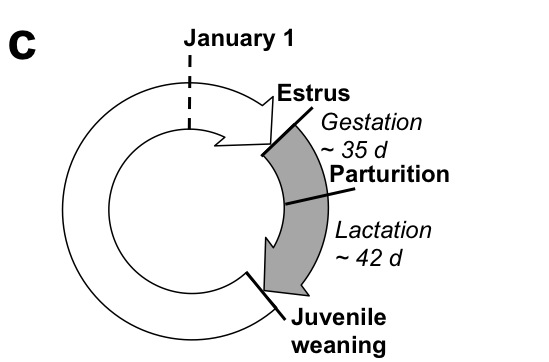
Among terrestrial vertebrates, the phenologies of mammals have been less studied relative to the rich diversity of work that has been conducted on birds. We have been monitoring the phenologies of Columbian ground squirrels and North American red squirrels (and have initiated new work on black-tailed prairie dogs) While ground squirrels and red squirrels are relatively similar in size (each < 1kg) and both occupy northern latitudes, they employ two very different approaches to survive the winter. Columbian ground squirrels hibernate for 8-9 months of the year and red squirrels remain active year-round, relying on a cache of hoarded conifer cones. Combined, they represent excellent models to better understand the causes and consequences of variation in mammalian phenologies. Current research questions include:
Representative Publications:
Among terrestrial vertebrates, the phenologies of mammals have been less studied relative to the rich diversity of work that has been conducted on birds. We have been monitoring the phenologies of Columbian ground squirrels and North American red squirrels (and have initiated new work on black-tailed prairie dogs) While ground squirrels and red squirrels are relatively similar in size (each < 1kg) and both occupy northern latitudes, they employ two very different approaches to survive the winter. Columbian ground squirrels hibernate for 8-9 months of the year and red squirrels remain active year-round, relying on a cache of hoarded conifer cones. Combined, they represent excellent models to better understand the causes and consequences of variation in mammalian phenologies. Current research questions include:
- How are traits at one point in the phenological sequence (e.g., entry into hibernation or initiation of food hoarding) related to traits at other points (e.g., emergence from hibernation or breeding date)?
- How does variation in phenological traits relate to variation in other life history components (e.g., litter size)?
- What are the relative roles played by phenotypic plasticity and microevolution in observed phenological shifts due to climate change?
- Are observed phenological shifts adaptive (i.e., sufficient to maintain population viability) in the face of climate change?
Representative Publications:
- Lane, J.E., L.E.B. Kruuk, A. Charmantier, J.O. Murie and F.S. Dobson. 2012. Delayed phenology and reduced fitness associated with climate change in a wild hibernator. Nature 489: 554-557.
- Boutin, S. and J.E. Lane. 2014. Climate change and mammals: evolutionary versus plastic responses. Evolutionary Applications 7: 29-41.
- Williams, C.T., J.E. Lane, M.M. Humphries, A.G. McAdam and S. Boutin. 2013. Reproductive phenology of a food-hoarding mast-seed consumer: resource- and density dependent benefits of early breeding in red squirrels. Oecologia 174: 777-788.